The first week of the MarineGEO Ola I ke kai biodiversity survey is complete, and what a week it’s been! All the various teams have come together: macro- and micro-invertebrates, fish collections, visual SCUBA surveys, algae, Autonomous Reef Monitoring Structures (ARMS), and microbes, to accomplish something truly exciting — as of this writing, our teams have identified at least three new species, and found a rare basal metazoan that is considered a “missing link” between unicellular and multicellular organisms. My team (visual survey) also identified 95 species of fish in Kāneʻohe Bay as well as several invertebrates.
So how did we do it? Well, as a member of the visual fish survey team, my typical day looked something like this…
5:30AM: Awaken in the Lanai Suites at the Hawai’i Institute of Marine Biology on the island of Moku o Lo’e to the sun just cresting over the Pacific Ocean, and the sound of the island’s many birds singing just outside my window. My habit of rising early on this trip might also be a symptom of lingering jet lag. Wander around the island for a bit to shake off the cobwebs.

View of the Bay in the morning from the Lanai Suites on Moku o Lo’e.

Critters wait to be sorted.

The camera rigs.

Zach packs the boat for the day.
6:15AM: Trek down to the kitchen and drink gallons of coffee, grab a bagel, and pack any snacks I’ll need for the day ahead. Watch sea cucumbers in the pond while I eat my breakfast.
7:00AM: Head up to the lab to check emails and peruse the fantastic diversity of organisms sitting in collections dishes brought up by yesterday’s efforts.
7:45AM: Get my SCUBA gear together and bring it to the dock, along with datasheets, transect tapes, and my underwater camera rig.
8:00AM: Jump on a boat (small whaler) with my dive buddy Zach and our HIMB driver, and motor out to one of the patch reefs that fill Kāneʻohe Bay. The bay has over 54 patch reefs, with the furthest being a 25 minute drive to the north from the HIMB.

Zach catches some rays on the way to our first site.

The transect tape laid out over the reef.

Emmett gingerly deploys a GoPro to monitor fish.
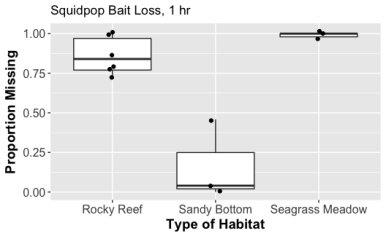
8:45AM: Anchor at our site and conduct our first Reef Life Survey (RLS) of the day. The surveys have typically been at a depth of about 3 meters (so shallow!), but the patch reefs quickly give way to rubble so we have stay closer to the surface to remain within the primary coral habitat. We run a 50 meter long transect, counting every fish we see and assigning it to a size class while swimming one direction, then turning around at the end and count small fishes that hide in cracks and crevices closer to the transect, as well as any large mobile invertebrates we see like shrimps, sea urchins, and sea cucumbers. All the while we try to take as many photos as we can so we can create an inventory of species with photo “vouchers”. Sometimes other researchers join us, often snorkeling above us as we work. The director of MarineGEO, Emmett Duffy (my boss), came out with us one day to deploy Squidpops and take GoPro video of the fish community. He’s hoping to relate the video he captured to the diversity that we observe on the RLS transects. At the end of every dive Zach and I take time to go on a fish photo safari at the surface. This is where many fish congregate and we’re hoping for that perfect shot that best showcases the characters of particular fish. To do that we need: a) good lighting, b) cooperative fish, and c) luck. We’ve had a little of “a”, a bit of “c”, and almost none of “b”. We’ve observed that the fish in Kāneʻohe Bay are rather skittish and shy of divers, particularly compared to many of the fish we encounter in the Caribbean that will swim right up to us. This makes photographing them a real challenge, but we’ve had a few success (with a little post-processing help from GIMP…)

Our fearless captain from HIMB takes us around the bay.

An ARMS deployed on the reef.

The CRAMP and RLS teams have a briefing before the surveys.

Sites ID’d for RLS. They are either historic CRAMP sites, or have ARMS deployed at them.
10:00AM: Haul anchor and move to the next reef on our hit-list. We tried to locate our dives where other work has been done previously, so we can compare and relate our data to existing or forthcoming datasets. Specifically, we did RLS on every reef that had ARMS deployed on it, as well as reefs where the CRAMP fish survey method has been done in the past for a total of ten reefs. Dr. Ku’ulei Rodgers has been spearheading the CRAMP surveys for many years and she was kind enough to let us tag along to show us her methods, and directly compare RLS to CRAMP. We’re hoping to use this method-calibration to relate the historic CRAMP surveys with other RLS surveys across the bay and beyond.
10:30AM: Conduct our second RLS survey and take lots of photos.
12:00PM: Return to Moku o Lo’e for lunch (sandwiches!) and catch up with the other researchers. The teams that are collecting organisms typically go out in the morning and afternoon to sample sediments and reef organisms. They use a range of techniques including: underwater vacuums to suck up small critters, collecting dead coral and breaking it open to find out what is hiding inside (lots of things), taking handfuls of algae and shaking the animals out of it, SCUBA collections, snorkel collections, plankton tows, and everything in between.
1:30PM: Get back on the boat to survey our final site of the day.
2:00PM: Last verse same as the first…conduct another RLS survey!
3:30PM: Return to Moku o Lo’e to rinse gear and grab another shot of coffee.
4:00PM: Begin the data entry and photo curation process. This is actually the most time-consuming part of visual surveys. We have to take our data sheets and enter them in a spreadsheet that will be brought together into the final database. We typically sit in the lab, surrounded by fish books, looking at the fuzzy photos (and one clear one) of the critters we saw, asking each other, “Hey, did you see that weird wrasse?” and “What the heck was that stripey fish?” We also gather together our photos and add one of each species to the photo library. As the week goes on, we replace subpar pictures of particular fish with better ones we captured that day. Our goal is to have at least one reasonably clear photo of each species that we saw. Sometimes we succeed, sometimes we don’t… All the while, other teams are swirling around us in the lab, handing specimens back and forth, looking at creepy-crawlies under the many microscopes, and jumping up and down when they find something new. When the computer screen starts to make my eyes water, I’ll take another walk between the tables to peek over the shoulders of the taxonomists. Crabs, fish, sponges, algae, seastars, and all manner of bizarre phyla sit in dishes, covering every bit of real estate on the tables.

Banded Damselfish (Abudefduf abdominalis)

White-spotted Puffer (Arothron hispidus)

Two-spot Goby (Asterropteryx semipunctatus)

Whitespotted Toby (Canthigaster jactator)

Oval Butterflyfish (Chaetodon

Hawai’ian Dascyllus (Dascyllus albisella)

Turkey Moray (Gymothorax meleaagris)

Moorish Idol (Zanclus cornutus)

Sailfin Tang (Zebrasoma velifer)
6:30PM: Break for dinner! We’ve had great catering this entire trip and everyone looks forward to the delicious meals that are served each night. We all gather together to eat, and assertively discuss and negotiate the use of boats for the following day. Limitations imposed by the numbers of boats, numbers of captains, and numbers of people wanting to go out are always a challenge, but we usually find collegial solutions to limited resources.
7:30PM: Back to the lab to sweat over fish photos for a few more hours (at least I hope it’s only for a few more hours).

Analysing the ARMS.

A crab collected from the reef.

Seastar in a dish.

Urchins and other organisms are sorted by species.

Burning the midnight taxonomic oil.
10:00PM: Go back to my room and fall asleep before my head even hits the pillow. Rinse. Repeat.
Now multiply that by six days and you have the beginning of an epic biodiversity inventory. The visual survey team has finished it’s job and we are leaving the island, but other teams will be going for another week to finish off their collections. After their data curation is complete, a process that can sometimes take months, everything they find will be uploaded to an open-access database. Our goal is to share everything we learn with anyone who wants to know.
It’s been a wild ride, I can’t wait to see how many more new species the MarineGEO teams find!